
Imagine having a microscopy system that can perform image-guided protein isolation automatically in a large scale, boosting efficiency and unlocking new possibilities for scientific discovery. As the scientific community strives to unravel the intricate details of the proteome, researchers find themselves at a critical juncture where scaling up is paramount due to the lack of a PCR-equivalent amplification method for proteins. Automation and AI are directly addressing the challenges that once stalled progress. By bringing forward new methods for isolating proteins in ultrahigh content with a subcellular precision and identifying them with mass spectrometry, these advancements are propelling us toward a future of discovering key protein constituents relevant to diseases or basic biology, all with an unprecedented level of precision.
Automation and AI in proteomics with Microscoop™
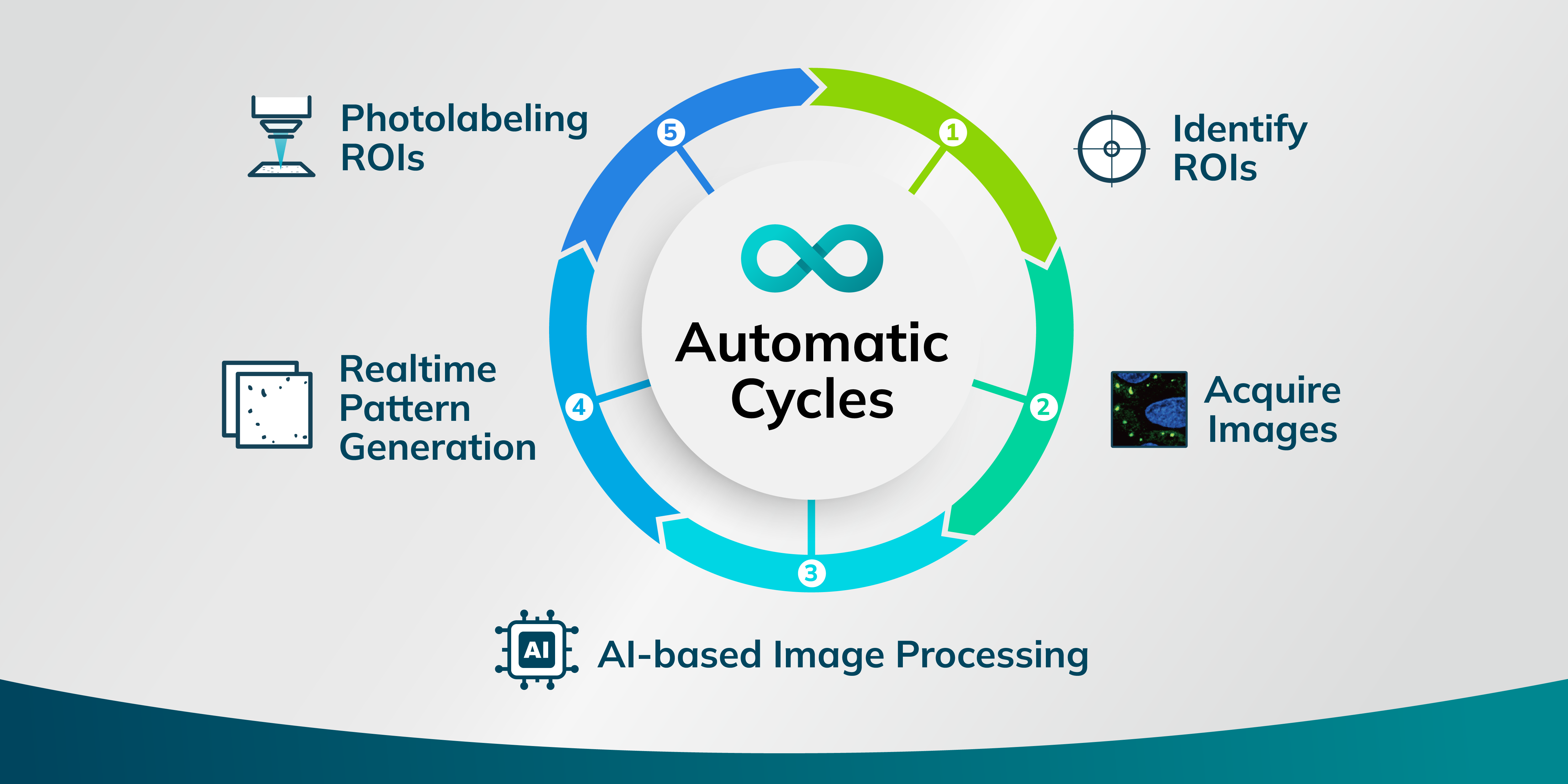
Syncell has turned the dream of automating the collection of high-quality subcellular samples for downstream analysis into reality, all with a simple press of a button. This achievement comes through the development of an advanced platform called Microscoop™. It integrates a computer-controlled microscope, image processing, and photo-biotinylation technology. Microscoop™ is equipped with a motorized epifluorescence microscope, a high-resolution sCMOS camera, and a two-photon light source, complemented by a specially developed photochemical probe. This allows for precise photo-biotinylation at the subcellular level within specific regions of interest (ROIs) in a sub-millisecond timeframe. Addressing the challenge of insufficient protein quantities from ROIs in a single field of view (FOV) for revealing submicron-sized targets, Microscoop™ efficiently performs automated targeted photo-biotinylation across ten thousand or more FOVs, ensuring an ample collection of biotinylated proteins for comprehensive proteomic analysis.
AI-driven recognition has transformed the way researchers analyze microscope images and identify ROIs. By incorporating AI-trained models, Microscoop™ enables real-time pattern recognition capabilities that are adept at identifying complex features. It excels at sorting through numerous FOVs that display similar characteristics, significantly enhancing the accuracy and speed with which ROIs are recognized. The automation of image-guided photo-biotinylation reduces the need for manual labor while ensuring uniform precision across experiments. This level of automation and precision is crucial in the context of subcellular protein studies, where identifying specific locations and interactions can provide critical insights.
A snapshot of a day with the Microscoop™
Researchers begin by finely adjusting the microscope to obtain sharp images of the pre-stained ROIs. Subsequently, they employ Syncell's segmentation software to precisely delineate areas of study, enhancing the clarity of the ROIs even in the presence of noisy or complex backgrounds. Once the labeling area is defined, the system automatically labels the entire predefined zone, processing each FOV in sequence. MicroscoopTM allows researchers to initiate an experiment, leave, and return the next day to a sample rich in biotinylated proteins targeted at the subcellular precision, ready for subsequent pulldown and mass spectrometry analysis.
Conclusion
The Syncell Microscoop™ offers distinct capabilities to advance spatial proteomics research. Its applicability to both cell-based and tissue-based samples makes it versatile for fundamental and clinical research domains. This shift empowers researchers to undertake proteomic studies on a scale that matches the complexity of biological systems, fostering discoveries that broaden our understanding of health and disease . These automation and AI models facilitate a comprehensive exploration of subcellular protein locations and interactions, providing insights indispensable for the advancement of biological research.
| Info@syncell.com | |
| 617-631-2746 | |
|
|
200 Dexter Ave, Watertown, MA 02472, USA |
|
|
|